Between 5% and 10% of all breast cancers are metastatic at diagnosis, and 20%-30% of patients diagnosed with early-stage breast cancer eventually progress to the metastatic stage. The most common type of metastatic tumor is estrogen receptor-positive (ER+) and human epidermal growth factor receptor 2-negative (HER2-), representing 68% of metastatic tumors.
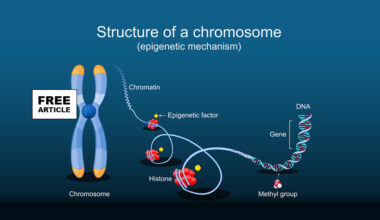
ESR1 is the gene that codes for estrogen receptor alpha (ERa) a protein that binds to the hormone estrogen and plays a key role in cell growth and development. Once activated by estrogen, the estrogen receptor alpha protein controls the activity of certain genes involved in cell growth, metabolism and reproductive functions. Mutations in the ESR1 gene are significant in breast cancer, where they can cause resistance to some hormone therapies and may lead to faster growth. These mutations are often acquired during treatment making them an area of focus for testing and treatment selection in advanced cancers.
ESR1 mutations are associated with resistance to endocrine therapy, the key treatment mode for metastatic hormone receptor-positive breast cancers. They are relatively rare in primary tumors, occurring in only around 1 in 100 cases, but may be found in between 10% and 50% of metastatic endocrine therapy-resistant tumors, suggesting that they play a key role in acquired treatment resistance and disease progression.
ESR1 mutations may affect tumor sensitivity to various therapies, both established and new treatments, hampering development of effective treatments. They have been associated with shorter progression-free survival and shorter distant recurrence-free survival.
In patients with metastatic ER+/HER2- breast cancer who show progression or recurrence during hormone therapy — with or without the addition of a CDK4/6 inhibitor (Ibrance, Kisqali or Verzenio) — Physicians recommend routine testing to detect acquired ESR1 mutations, for example through liquid biopsy, to guide subsequent targeted therapy decisions.
Newly approved Inluriyo is monotherapy for the treatment of adult patients with ER+/HER2-, locally advanced or metastatic breast cancer with an activating ESR1-mutation, who have disease progression following treatment with an endocrine based regimen. In pre- or perimenopausal women and in men, Inluriyo should be combined with a luteinizing hormone-releasing hormone agonist.
The drug’s active ingredient, imlunestrant, is an estrogen endocrine agonist that antagonizes and degrades wild-type and mutant estrogen receptor alpha, leading to inhibition of estrogen receptor-dependent gene transcription and cellular proliferation in ER+ breast cancer cells.
A phase 3, randomized, open-label study showed that imlunestrant as monotherapy improved progression-free survival compared with fulvestrant, a selective estrogen receptor degrader or exemestane, a steroidal aromatase inhibitor in adults with ER +/ HER2 ‑ locally advanced or metastatic breast cancer with an activating ESR1-mutation when they had disease progression following treatment with an endocrine-based regimen.